There are four accreditation bodies (ABs) operating in Thailand however, only the National Standardization Council (NSC) – Thai Industrial Standards Institute (TISI) – Ministry of Industry is a full member signatory to both the ILAC MRA and IAF MLA. The Bureau of Laboratory Quality Standards (BLQS-DMSc) – Department of Medical Sciences – Ministry of Public Health, and the Bureau of Laboratory Accreditation (BLA-DSS) – Department of Science Service – Ministry of Higher Education, Science, Research and Innovation hold full membership and signatory status to only the ILAC MRA while the Office of Standard Accreditation (OSA) – National Bureau of Agricultural Commodity and Food Standards (ACFS) – Ministry of Agriculture and Cooperatives holds full membership and signatory status to only the IAF MLA. All of these ABs are Government Agencies working as the agency-in-charge for standardization in a specific field of accreditation (such as testing and calibration laboratories, medical laboratories, reference material producers (RMPs), and product certification programs). Their specific scope of activity relates to the National Standardization Act B.E. 2551 (2008) of the national standard committee under the Cabinet approval.
The implementation of the United Nations Sustainable Development Goals (SDGs), the benefits of the multilateral MRA for greater efficiency, cost savings on the ease of doing business policy, and the ongoing global pandemic of the coronavirus disease 2019 (COVID-19) provide significant opportunity for the Thai Public Policy Government to improve its own performance. A Memorandum of Understanding (MoU) on a single platform for accreditation was signed between the four ABs to strengthen the cooperation on 15th December 2020. This partnership aims to reduce the redundancy and unnecessity of multiple onsite assessments and accreditations from different ABs with specific areas of accreditation activities for customers.
Presently, the unified accreditation reform has commenced with testing laboratory accreditation by using joint assessments between ABs under the ‘single scheme’ of the same standard (i.e. ISO/IEC 17025) and under the ‘multiple schemes’ of different standards (i.e. ISO/IEC 17025 and ISO 15189). Currently a laboratory can apply with only one AB for any scope of testing where that AB has the MRA recognition. For example, currently, a laboratory has to apply for ISO/IEC 17025 testing scope of accreditation with NSC for industrial chemicals; with BLQS-DMSc for pharmaceuticals; and with BLA-DSS for waste water. From now on, the laboratory will be able to apply to just one AB as a single point of contact (“one stop service”), for the entire scope of testing activities, and still be verified compliant with the standard. The platform however, is currently only voluntary, so a customer may maintain accreditation with each individual AB as per usual should they wish.
The next step, is to set up ongoing monitoring and reporting systems that interface with this platform. We look forward to working with the simplified and harmonized processes and procedures of each AB to expand the unified accreditation model., In the future we plan to establish the “one application – one platform – one certificate” for testing laboratory accreditation, and also expand this platform to other MRA/MLA scopes such as reference material producers (RMP), proficiency testing providers (PTP), management system (MS) as well as extending the scope of the recognition to the MRA/MLAs to cover other conformity assessment body activities in Thailand (e.g. biobanking facilities specifying accreditation complying with ISO 20387 and medical device accreditation complying with ISO 13485).
We held a public hearing addressing these proposed changes on Wednesday, September 1, 2021 via the zoom cloud meeting application to communicate and promote the changes to our customers, and also to receive input and feedback. Approximately 650 participants shared their thoughts with discussion and opinions regarding this joint initiative. This input will allow us to pursue a better modifying process. Participants at the public hearing approved of the initiative and few of the customers have already decided to apply using this platform on their next cycle of assessment supporting sustainability in accreditation. Finally, this joint supporting service was approved to start in October 2021 and is expected to be a great success with acceptance from customers. It will also help to stimulate competitiveness in the economy, and keep up with global changes especially in this time of COVID-19 restrictions.


On 28-30 September Minsk, Belarus, hosted The Belarusian Industrial and Innovation Forum 2021 – one of the largest international events in the field of industry, high technology, innovation and scientific and technical development. The purpose of the exhibition is to promote innovative industrial equipment, products and technologies of domestic and foreign companies to regional and international markets and to strengthen business contacts and share best practices.
Within this Forum on September 29, 2021 the Belarusian State Centre for Accreditation held a breakout session covering topical issues of accreditation.
The benefits of international recognition of accreditation for governors, industry, consumers was discussed as well as the value of the ILAC MRA and IAF MLA marks on conformity assessment documents for business and industry. BSCA also took the opportunity to draw more attention to the global data base IAF CertSearch as an international online platform to provide the validation of accredited certification worldwide.


Cofrac* is used to organising events for its customers of its different divisions. Usually offered face-to-face, theses meetings have been reinvented due to the pandemic.
Following two “Accreditation and Inspection meetings” organised in 2016 and 2018, Cofrac offered a completely redesigned event in June 2021, to accredited bodies and those interested in accreditation for inspection activities.
With a studio worthy of a television production and several projectors and cameras pointed at concentrated speakers, flawless animation, discussions, presentations and videos broadcast and available for streaming; the organisation of this event was undoubtedly a great first and success for Cofrac!
Called “Accreditation in the inspection field: let’s talk about it!”, this event took place remotely, filmed and broadcast live from Paris. Only the members of the Inspection division were present on D-day in the studio to encourage their colleagues in this unprecedented exercise.
On the program for this half-day: updates and projects from the division, a presentation of the most frequent findings observed during the assessments as well as a point on the associated requirements, and a focus on the document GEN REF 11 – General rules for reference to accreditation and to international recognition arrangements – which gave some clarification on how to use the reference to accreditation.
The event was punctuated by live surveys, to collect participants’ reactions, and video interludes, one on the Cofrac digitisation project, and a second one explaining the different types of independence in the inspection field.
The organisation of this remote meeting met the expectations of the 142 people who participated via the streaming platform. Indeed, out of the 77 who answered the satisfaction questionnaire, 99% said they were satisfied and very satisfied. When asked “Did you like the TV set format?“, the answer was unanimous: 99% yes! Further proof of the success of this event is that 73% of respondents said they would prefer this remote format for a future event!

* The French accreditation committee (Cofrac).
EIAC launched a number of new accreditation schemes:
Crime scene investigations
The Emirates International Accreditation Centre (EIAC) has launched its ISO/IEC 17020:2012 accreditation scheme for inspection of crime scenes. This is a first of its kind in the region. EIAC is already running ISO/IEC 17025 accreditation schemes for forensic and digital forensic testing laboratories. This new accreditation scheme will enhance the confidence in the reliability and accuracy of data produced during criminal investigations.
Medical sample collection centers and laboratories
This scheme is based on ISO/TS 20658: Medical laboratories- Requirements for collection, transport, receipt, and handling of samples and covers the essential and very important aspect of medical testing. Ensuring the integrity of the samples is crucial reliable testing.
Inspection for clean room and biosafety cabinet
This scheme is based on ISO/IEC 17020:2012 and provides reliable tools to ensure the better environmental monitoring.
EIAC accredited first day care surgery center
EIAC has accredited the first Day Care Surgery Centre in the region. The scheme is based on the requirements of the International Society for Quality in Health Care (ISQua). This scheme was developed to address the growing need for quality management in the healthcare sector.
EIAC signed MoU with Dubai Health Authority
EIAC has revived its Memorandum of Understanding (MoU) with Dubai Health Authority (DHA). A delegation from DHA visited the offices of EIAC and the progress on the MoU was reviewed and it was resigned. The DHA is an important stakeholder for EIAC in its accreditation services. DHA has recognized EIAC as one of the accreditation service providers for healthcare sector. The EIAC extended its services to cope with the challenges brought about by the COVID-19 pandemic and accredited conformity assessment bodies for COVID-19 testing and personal protection equipment testing. The EIAC has accredited 168 medical laboratories and one day care surgery center.

EIAC CEO with visiting DHA delegation
EIAC participating in regional and international events
As a major accreditation body in the region, EIAC actively participates in regional and international activities.
Ms. Amina Ahmed Mohammed, Chief Executive Officer of EIAC, attended the 8th International Halal Food Conference on 1st September 2021. She presented on the “Role of EIAC Accredited Halal Conformity Assessment Bodies in Developing Halal Trade”. Mr. Mohammad Abdelmotagaly, Advisor of EIAC, gave a presentation on the “Approach Towards Harmonizing Halal Standards”.
Ms. Amina Ahmed Mohammed also attended the symposium of Saudi Accreditation Council. The symposium celebrated World Accreditation Day. Ms. Amina spoke on the role of Arab Accreditation Cooperation in the region for supporting the accreditation bodies.

EIAC CEO addressing the International Halal Food Conference
EIAC’s accreditations helping business entities and decision makers
The United Arab Emirates has a strong quality culture. The EIAC is playing a vital role in maintaining the highest levels of quality in various fields. Business entities, and public and private decision makers are widely accepting the accreditation granted by EIAC.
An EIAC accredited inspection body was contracted to inspect all stages of the world’s tallest Ferris wheel known as “Ain Dubai”. It is 250 meters (820 ft) tall and is due to open on 21st October 2021. The EIAC accredited inspection body has certified the whole journey of the Ferris wheel from reviewing the design, to inspecting the manufacturing process and the functionality, and verifying the safety of the wheel post installation.
EIAC Director of Certification Bodies Accreditation Department and the Advisor of EIAC visited Dubai airport and immigration authorities and discussed persons certifications for various fields related to airport and immigration operations.

EIAC representative’s briefing to Dubai airport authorities
EIAC’s peer evaluation by ARAC
The periodic peer evaluation of EIAC was conducted on 23-27 May 2021. The scope included ISO/IEC 17025, ISO 15189 and ISO/IEC 17020 for testing, calibration, medical laboratories and inspection bodies, and the evaluation was conducted remotely.
EIAC’s remote peer evaluation

ISRAC: A remote/on-site hybrid for accrediting PCR based COVID-19 molecular detection
The Coronavirus crisis forced ISRAC to adapt quickly. As in all other affected economies, performing an on-site assessment presented a serious health risk to both the laboratory and the assessment staff. The risk was highest when assessing the performance of medical laboratories, especially those seeking accreditation for routine PCR-based COVID-19 detection.
In Israel, there is no regulatory demand for ISO 15189 accreditation. Nevertheless, two laboratories sought accreditation for a kit-based system.
Some of the challenges were instantly apparent. The technical assessor, while having vast knowledge needed to adequately assess the activity, was in a risk group. Furthermore, vaccines were not available.
To solve this challenge, aw remote assessment was used. However, this concept was relatively new. As e could not fully rely on remote tools, as it was unclear how efficient such an assessment can be. For that reason, we developed a risk-based approach to the viability of such an option. Including proper supporting infrastructure, type of assessment, and limitations to conducting observations and interviews properly. The last parameter in this risk-based approach was the presence of an ISRAC representative on-site, as we believed such a presence would lower the risk significantly.
Accordingly, the laboratory representatives assured us beforehand that the Wi-Fi signal is strong in all relevant workspaces and a lead assessor with experience in molecular biology was on site, along with a laptop setup with a remote communication program.
The assessment was certainly adequate, in part thanks to the cooperation of the staff. However, several problems arose. The two main issues revolved around the equipment used for the assessment. The noise in the room made the communication hard. Furthermore, the camera quality prevented the technical assessor from seeing items in a proper resolution, and she often relied on verbal descriptions given by the assessor on site. This dynamic slowed the process significantly, and arguably, without a representative on-site the assessment could not have been carried out adequately.
To improve on this performance, the equipment used at the second laboratory was upgraded and included Speakers, a microphone, and a Go-pro camera. The lab provided a cart that acted as a sort of portable assessment station, moving from one bench to another, and providing the technical assessor with much more information than before. The infrastructure needed was more of a challenge in this case, as the lab is set up underground, and the relevant workspaces were more spaced out. However, clear communication between ISRAC staff, lab management, and the hospital’s IT staff resulted in a clear and strong signal throughout the day. The addition of the go-pro camera allowed for greater flexibility in the visual aspect of the assessment. it was possible, for example, to examine tip positioning in the robot, hard-to-reach calibration stickers, and proper use of equipment inside the biological cabinets. The fact that the same assessors, both the technical and the lead, were the same in both laboratories was also beneficial as even this limited experience with a hybrid assessment made the second one better.
A pandemic made it necessary to come up with a new tool that would allow us to continue with our duties. While unforeseen issues had to be overcome, constant improvement and risk management allowed ISRAC to establish an adequate tool for remote and hybrid assessments. The assessors stayed safe, and all the aspects of the ISO 15189 standard were adequately examined. Hopefully, this new tool will not be put aside even after a global return to normalcy.


NABL has taken up a unique initiative to promote the services of accredited Proficiency Testing Providers (PTPs) and Reference Material Producers (RMPs) and sensitize laboratories on the available infrastructure and services.
This year 5th PTP/RMP conclave was conducted on 30th – 31st August 2021 by NABL virtually which saw participation from not just the local but the international community including China, Iran, Mexico, Sri Lanka, etc. Chief Guest, Prof. HE Ping, CNAS, APAC PT Chair, provided an update on the revision of ISO/IEC 17043.
The PTP/RMP conclave also saw presentations from PTPs and RMPs on their PT plans and RM productions. The presentations are then judged by NABL experts based on technical aspects, content, presentation time, etc. and prizes were given to the top three PTPs/RMPs of the 11 presentations in the conclave.
For the full article on 5th PTP/RMP conclave please visit the NABL Newsletter August 2021 (Link: https://nabl-india.org/wp-content/uploads/2021/09/NABL-Newsletter-Aug-2021.pdf)
Note:
National Accreditation Board for Testing and Calibration Laboratories (NABL) is a Constituent Board of Quality Council of India (QCI), an autonomous body under Department for Promotion of Industry and Internal Trade (DPIIT), Ministry of Commerce and Industries, Government of India.
NABL is an accreditation body providing accreditation services to CABs:
*CABs accredited as on 30 September 2021

NATA pushes for accredited travel testing for COVID-19 to meet global standards
With Australia’s international borders soon to re-open, NATA has been actively urging the Australian Federal Government, aviation companies and international travel agencies to mandate use of accredited COVID-19 testing organisations and procedures to provide travellers assurance and confidence the tests conform to globally recognised standards.
While vaccination rates are rising around the world, vaccinated persons can still carry COVID so the need for diligent testing for both arriving and departing international travellers will continue to be a focus.
NATA believes that any laboratory performing COVID-19 tests should not only be accredited, but also be recognised under the International Laboratory Accreditation Cooperation (ILAC) framework.
In Australia, NATA has already accredited more than 160 facilities for COVID-19 tests including polymerase chain reaction (PCR) and more recently, Rapid Antigen Testing (RAT).
NATA firmly believes that specifying accreditation for COVID-19 is the only way for regulators to ensure the reliability of tests and to help reduce the impact on health, trade and the Australian economy.
——————–
View NATA CEO, Jennifer Evans, discussing travel testing with the BBC.
View NATA’s official Media Release
For additional information, please contact Brendon.moo@nata.com.au or Liam@commswork.com.au
Why NATA? A new video series
“You can’t be what you can’t see” is often used as a motto to encourage diversity.
NATA, Australia’s leading accreditation authority, is using “they say you can’t be what you can’t see” as a vision to deliver a new video series that brings visibility to the NATA brand and showcases and promotes the benefits of accreditation.
Titled ‘Why NATA?’, the series uses video storytelling to make an emotional connection by highlighting what NATA does and the role it plays in helping consumers, in every aspect of their daily lives – from the water we drink, the food we eat, the houses we live in or the cars we drive.
NATA produced the first episode as a “proof of concept” which you can see https://bit.ly/3pcBWNK
The spot features a family going about their daily routine and highlights how NATA impacts their lives and how standards exist all around them.
NATA is now planning to produce a series of episodes which will touch upon the many industries NATA works with, from agribusiness, animal health, the environment and human pathology, to forensic science and sleep disorders.
For additional information, please contact Brendon.moo@nata.com.au

To be is as essential as to look and at the National Accreditation Body of Colombia – ONAC, we are convinced that the evolution we have been cultivating for some time now and our willingness to change for the better must be reflected not only in our actions, but also in our “look and feel”; in that important part that connects with all our audiences.
Therefore, thinking about ONAC’s purpose as a permanent promoter of accreditation, understood as a fundamental activity for a sustainable economic development supporting and strengthening the country’s quality infrastructure, we carried out a review process of all aspects of such an organization. We contrasted it against our values and evolution during the last years, leading to a comprehensive redesign of our brand, achieving greater alignment with our strategic objectives, our organizational evolution, and the promise of connecting the quality of Colombia with the rest of the world in order to increasingly help position the country´s image and enhance its international recognition. ONAC is an ally inviting all its stakeholders to continue consolidating it as a Hub Catalyst of the world of quality that generates trust, through inspiring and convening leadership.
As a result of this evolution, the new logo that identifies the National Accreditation Body of Colombia was redesigned, and with it, the new Accredited and Associate ONAC Symbols, as shown below. Additionally, a new accreditation certificate was designed to reflect the new brand, which is already being used.
ONAC’s Logo

Accredited Symbol and Associate Symbol


Accreditation Certificate


The National Accreditation System of the Republic of Uzbekistan has had several developments since the accreditation of conformity assessment bodies was first introduced in 2013 with the adoption of the Law on Conformity Assessment.
In 2014, the first accreditation program for testing laboratories was introduced by the Uzbek Agency for Standardization, Metrology and Certification in accordance with the requirements of ISO/IEC 17025. In 2014/2015 the accreditation system was extended to inspection and certification bodies of management systems, persons, and products. Institutional reforms in 2017/2018 separated accreditation from other conformity assessment activities. The Center for Accreditation (O’ZAKK) re-established in 2018 and to-date has accredited over 300 conformity assessment bodies.
It should be noted that O’ZAKK’s accreditation services have been fundamentally transformed to better serve interest groups, ensure confidence in the market and demonstrate competence by providing access to the latest information about accredited organizations on the website – www.akkred.uz.
The O’ZAKK team of 55 employees, and pool of 46 assessors and 100 technical experts has been able to expand its presence in the international community through co-operation with the following accreditation bodies: TURKAK (Turkey), ENAS (Emirates), NCA (Kazakhstan), KAB (Korea), Rusaccreditation (Russia) and PCA (Poland).
Another important step for the Uzbek National Quality Infrastructure is the country’s first internationally recognized calibration service, when TURKAK officially accredited the National Metrology Institute of Uzbekistan (O’zMMI) on 22 March 2021 for selected calibrations in the areas of mass, scales, pressure and temperature.
As of February 2018 O’ZAKK is an associate member of APAC. O’ZAKK joined ILAC in March 2018 as an affiliate member and its membership was successfully upgraded to an associate status in April 2020. And in December 2020 O’ZAKK was successfully granted membership status to the IAF. Moreover, O’ZAKK’s experts represent Uzbekistan in ISO/CASCO’s conformity assessment standards development activities and participate as a member of ILAC’s Arrangement Committee (ARC).
O’ZAKK continues its development and will apply for APAC full membership status to become an APAC MRA signatory in 2021 with plans to achieve IAF MLA and ILAC MRA signatory status in 2022.




Standards have been widely used as government and regulators are constantly called upon to make decisions related to the health, environment, and welfare of consumers which may entail the development or update of regulatory and legal requirements.
Similarly, the Maritime Industry Authority (MARINA) of the Philippines has released the MARINA CIRCULAR NO. 2018-02 stipulating that, “All MARINA-Registered Shipyards licensed under this Circular shall be required to secure ISO 9001:2015 on Quality Management System and its latest amendments from a certification body accredited by the Philippine Accreditation Bureau (PAB).”
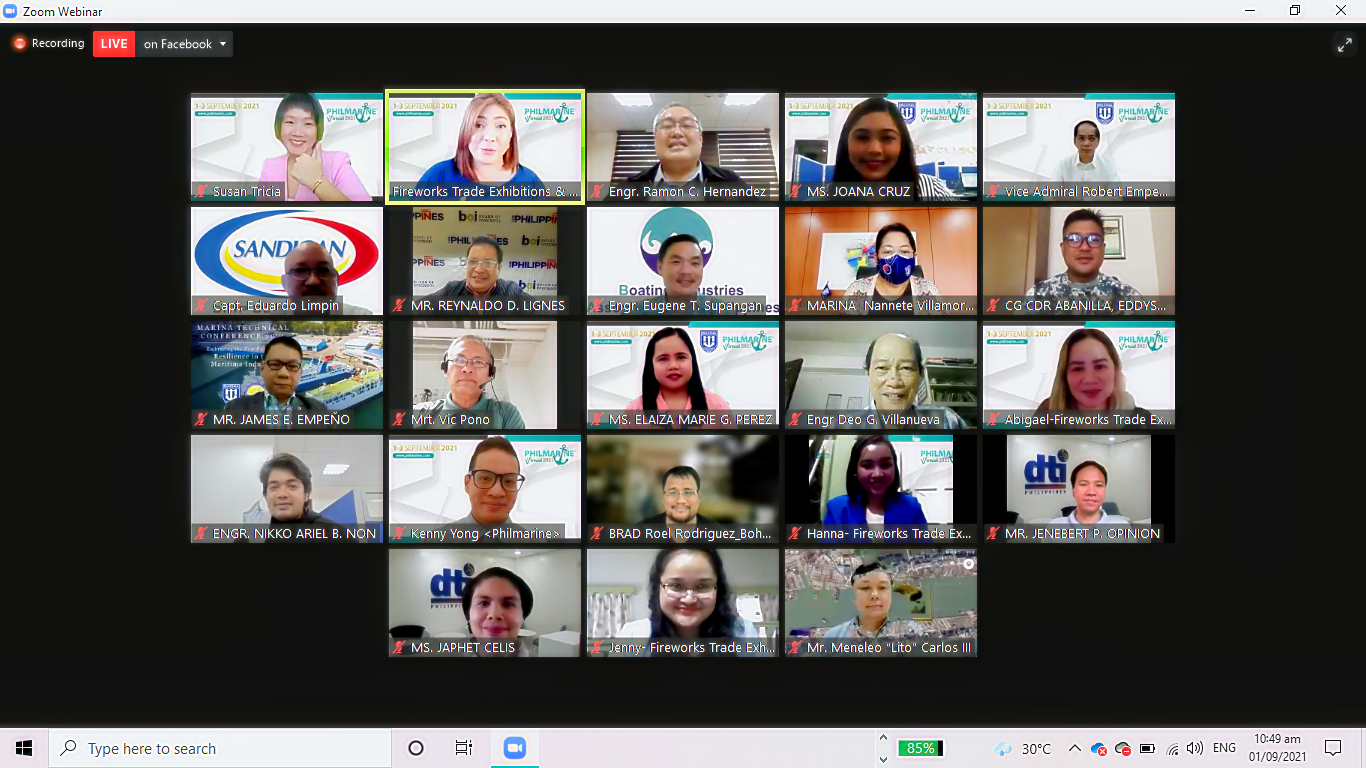
Many of us may ponder upon the relevance of ISO 9001 in the Shipbuilding and Ship Repair (SBSR) sector. This question was answered when Engr. James E. Empeño, Director of the Philippine Accreditation Bureau (PAB) was invited by MARINA on 01 September 2021 to discuss the significance of quality management systems in this highly-specialized industry.
During the 7th Philippine Marine 2021 Virtual Expo’s SBSR Technical Conference, Dir. Empeño explained that ISO 9001 is a standard that sets out the requirements for a quality management system. It helps businesses and organizations to be highly efficient and more customer-focused.
“ISO 9001 is suitable for organizations of all types, sizes, and sectors. One of the key improvements of ISO 9001:2015 was to make it more applicable and accessible to all types of enterprises,” said Dir. Empeño.
A well-implemented quality management system helps organizations to clearly define their objectives and identify business opportunities while making their customers a top priority. Apart from these benefits, it also aids businesses in ensuring that works are done efficiently, statutory requirements are met, organization-associated-risks are addressed, and markets are expanded because of the use of internationally-accepted standard.
Dir. Empeño’s discussion included some useful tips that would guide organizations in getting started with the implementation of ISO 9001. He also presented some success stories in adopting the said standard to provide further inspiration to those who are yet to pursue certification.
The demand for the certification in the SBSR and other sectors is expected to increase in the coming years due to the continual push of some regulators to conform to international standards.
As of writing, there are already 12 shipyards certified to ISO 9001:2015 apart from the 58 SBSR facilities with ongoing applications for ISO certification in the Philippines.
According to MARINA, the 7th Edition of Philippines Marine (PHILMARINE) 2021 is the only specialized maritime, shipbuilding, and offshore event in the Philippines that brings together an international congregation of maritime, shipbuilding, offshore, and their supporting industries to showcase the latest developments in the maritime industry. It aims to improve the current shipbuilding technology and equipment to help maintain the Philippines’ current status as the world’s fifth-largest shipbuilding industry.