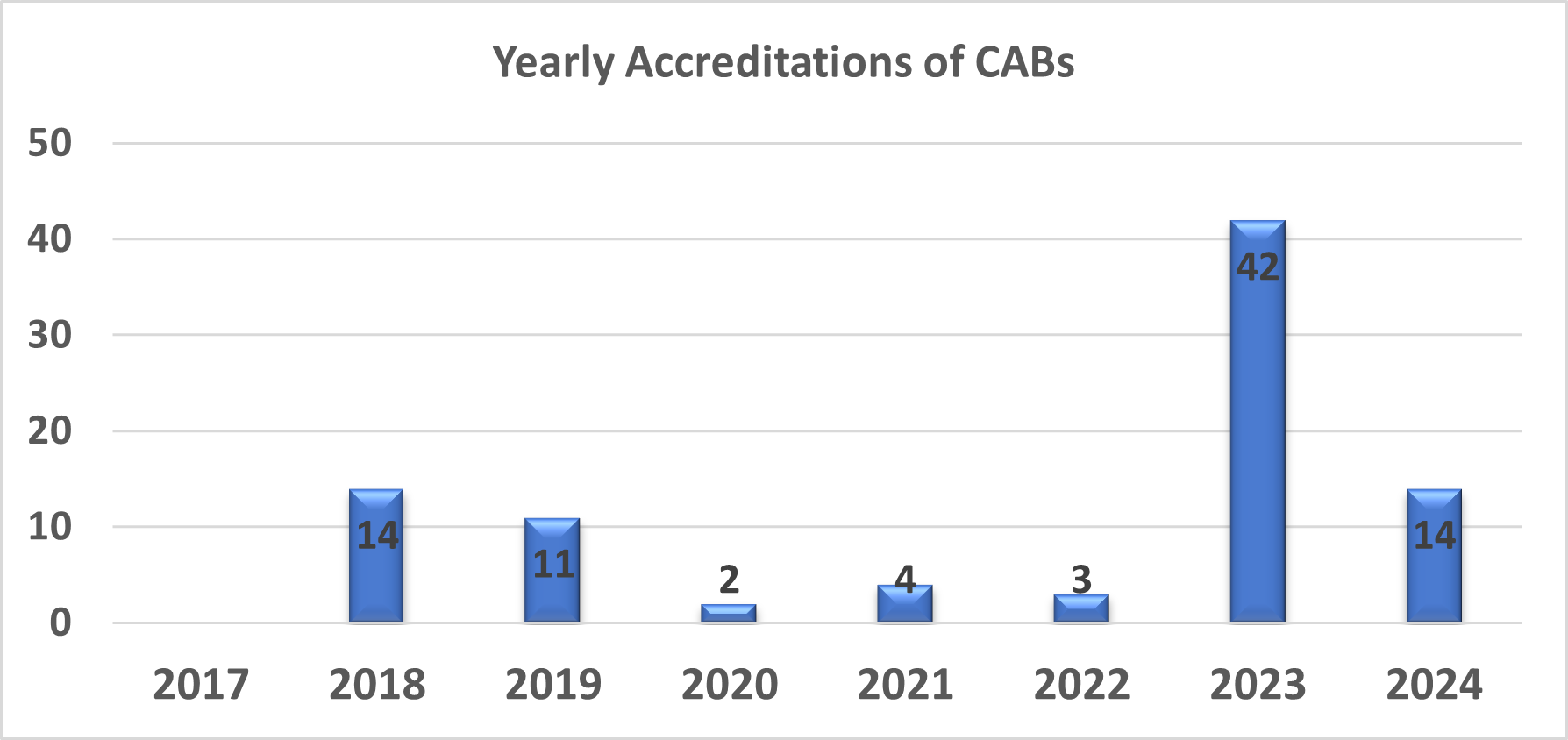
The QAI Centre for international Accreditation (QAI CIA), a fairly young organisation, started its accreditation activities for Conformity Assessment Bodies (CABs) in 2017 with the following accreditation programmes:
2017
• Accreditation of Medical Testing Laboratories as per ISO 15189
2018
• Accreditation of Testing laboratories as per ISO/ IEC 17025
2021
• Biobanking Accreditation as per ISO 20387 (For the First time in India)
2022
• Accreditation of Calibration laboratories as per ISO/ IEC 17025
2023
• Accreditation of Proficiency Testing Providers as per ISO 17043
• Accreditation of Reference Material Producers as per ISO 17034
• Accreditation of Inspection Bodies as per ISO 17020
We are happy to announce that QAI CIA achieved international recognition by joining the APAC MRA for Testing (ISO/IEC 17025) and Medical Testing (ISO 15189) on 31 October 2022 and the ILAC MRA for Testing (ISO/IEC 17025) and Medical Testing (ISO 15189) on 10 December 2022. This makes us the second AB in India to achieve membership of the ILAC MRA. We intend to add more accreditation programmes to the ILAC MRA as we progress.
This recognition has helped us in expanding and promoting our business, and accepting a greater number of applications for accreditation.
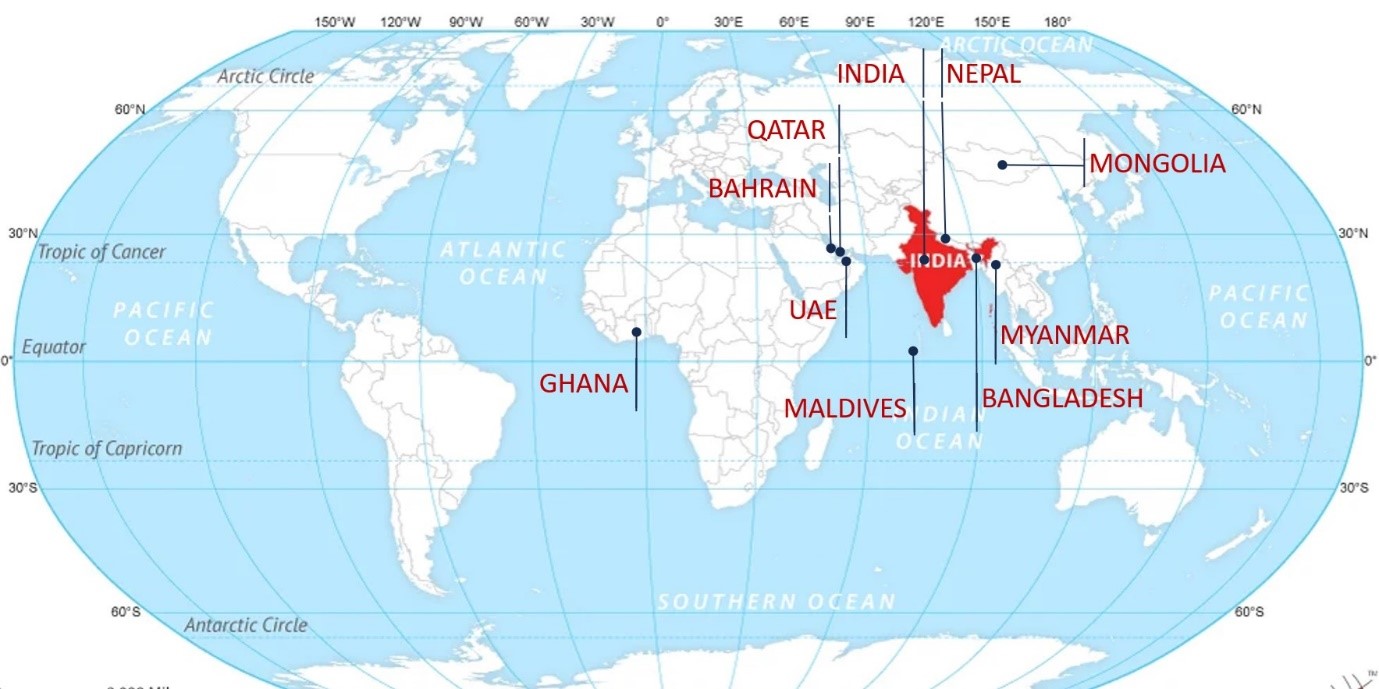
We are also pleased to announce that QAI CIA is now present in Asia, Africa and the Middle, East as an accreditation partner. We are motivated to excel in supporting countries that may not have their own Accreditation Body and / or a specific accreditation programme.
We are committed to hold to our Vision and Values by providing a credible accreditation mechanism. We are certainly would like to contribute towards our national government’s agenda of Viksit Bharat (Developed Bharat) in the conformity assessment ecosystem and do our bit to take Bharat (India) to the 1st World Ranking in Quality and Accreditation Infrastructure.
Further, we are open for bilateral collaboration to learn, share and support these shared goals with organisations in different countries.